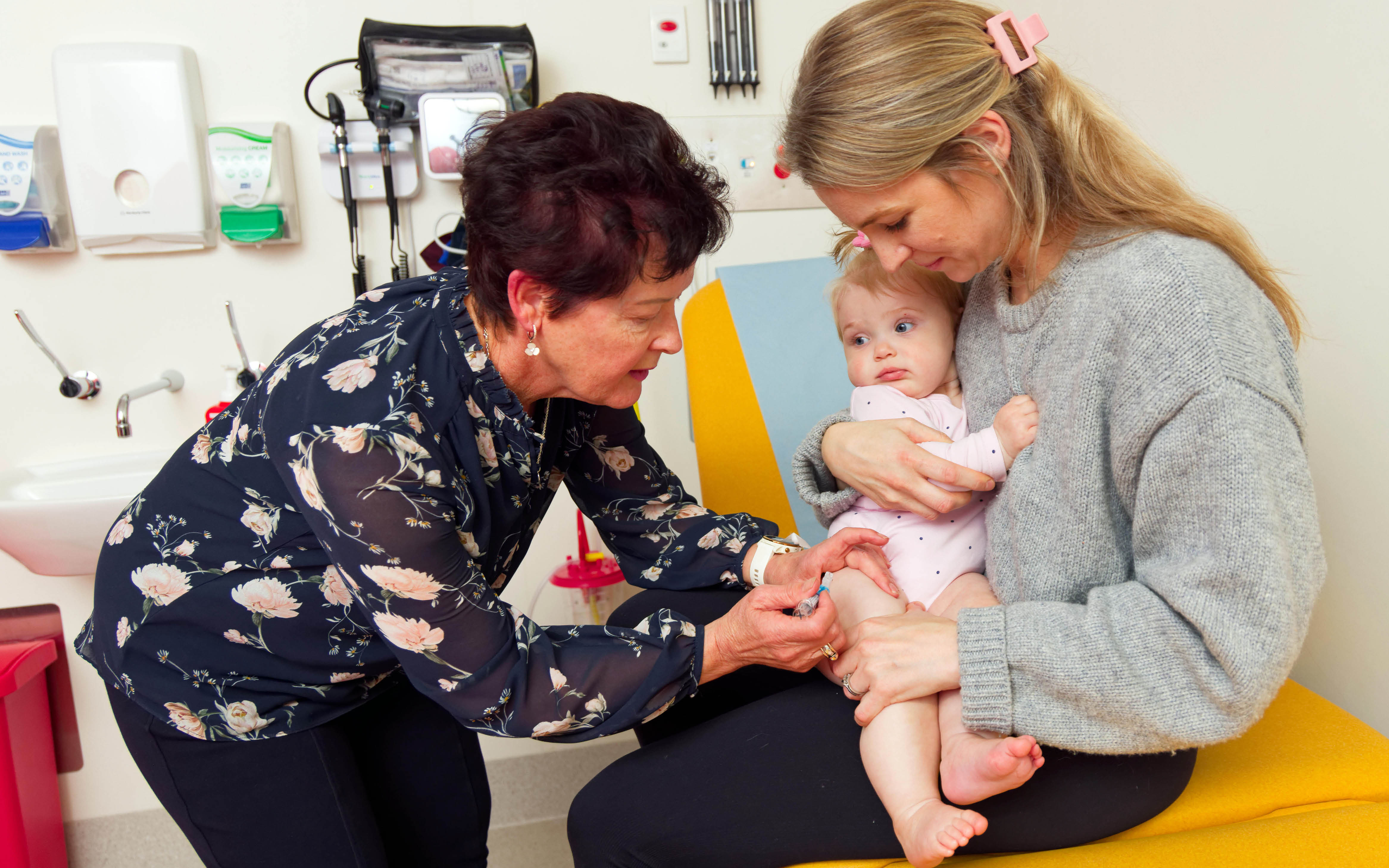
The Vaccine Trials Group (VTG) team of doctors, nurses, researchers, scientists, students and phlebotomists is testing and evaluating the safety and effectiveness of new vaccines for a range of diseases, as well as monitoring vaccines.
The VTG was established in 2000 to provide a coordinated approach to the development, delivery, assessment and promotion of vaccines, infectious disease and allergy treatments in the community. The team plays a vital role in the development and evaluation of vaccines recommended for children and adults throughout Australia.
The VTG leads national studies focusing on a range of diseases including influenza,
pneumococcal, meningococcal, respiratory syncytial virus (RSV) and whooping cough.
Team leader

MBBS MRCP(UK) FRACP
Head, Vaccine Trials Group
Team members (18)
Clinical Research Manager

BSc BMedSc(hons) PhD
Senior Research Fellow

Zoe Ellis
Clinical Laboratory Coordinator

Camille Gibson
Research Nurse

Jan Jones
Laboratory Research Assistant

Karen Jones
Executive Assistant

Sonia McAlister
Postdoctorial Researcher

Kieran Veale
Research Nurse

Ushma Wadia
Clinical Research Fellow

Kerrie Lisgaris
Research Nurse

Jonah Macliver
Laboratory Research Assistant

Shania Tansil
Laboratory Research Assistant

Chantalia Tedja
Laboratory Research Assistant

Madeline Ong
Clinical Research Fellow

Julia Kets
Research Nurse

Louise Ghosh
Research Nurse

Matthew Harms
Laboratory Research Assistant

Edison Foo
Laboratory Research Assistant; PhD candidate
Vaccine Trials Group projects
Featured projects
ATOMIC Ears: A Phase IIB randomised controlled trial to assess safety, tolerability and acceptability of a 5-day Dornase alfa treatment as an adjunct therapy to ventilation tube insertion for otitis media in children
Paediatric Active Enhanced Diseases Surveillance (PAEDS)
PAEDS monitors for key vaccine preventable conditions and severe side effects from vaccine in 5 paediatric hospitals in Australia.
Other projects